Contents
Introduction
Atoms have discrete energy states as a result of the unique set of energy levels their electrons can occupy.
In this post, we will explore atomic energy levels and see how they give rise to the characteristic emission and absorption spectra of the elements.
Let’s begin!
Discrete energy levels in atoms
Atoms consist of a positive nucleus made of protons and neutrons surrounded by electrons which orbit the nucleus. The atom is held together by the electrostatic attraction between the positive nucleus and the negative electrons.
However, the electrons do not “orbit” the nucleus in the classical sense. Rather, they occupy discrete orbits or energy levels, with the lowest energy level — called the ground state — being the most stable.
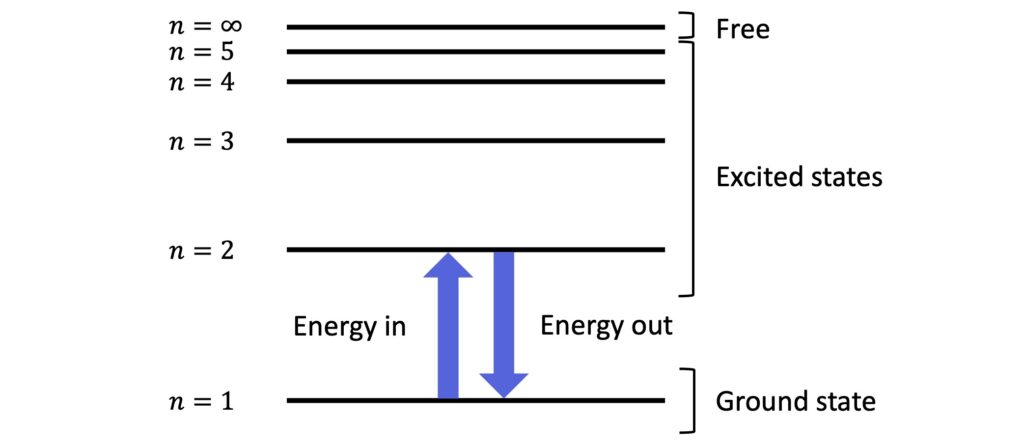
An electron can transition up to a higher energy level if it is given the right amount of energy. When it occupies a higher energy level, it is said to be excited!
In excited energy states, the atom is less stable and after a time the electron spontaneously falls back down to a lower level, releasing a photon in the process. The energy of the photon, \(hf\), is equal to the difference in energy between the two energy levels of the transition:
\(\Delta E =E_2-E_1=hf\)
Atomic energy levels are negative
Atomic energy levels are numbered from \(n=1\) (the ground state) to \(n=\infty\), which represents a free electron which has been completely removed from the atom. Removing an electron from an atom is called ionisation because atoms with missing or extra electrons are called ions.
The fully removed, free electron is no longer subject to the attraction of the positive nucleus. It therefore has zero electrostatic energy. But remember, we have to put energy in to remove the electron. As a result, the process of removing the electron increases the electron’s energy to zero. Energy levels in atoms are therefore negative!
For example, here are the values of the first five energy levels of hydrogen:
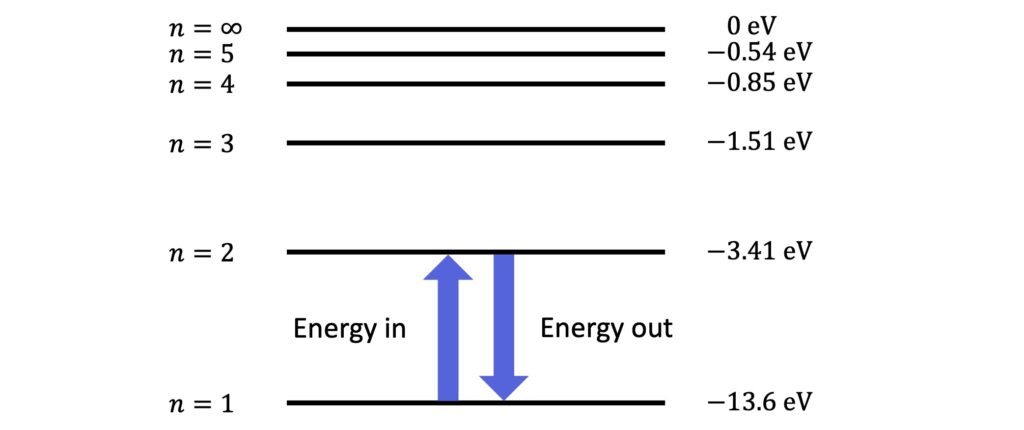
These values belong uniquely to hydrogen. Indeed, every element has its own characteristic set of energy levels. For example, the energy levels of hydrogen are different from those of helium.
The values are also very tiny, and are typically given in units of the electron volt (\(\rm{eV}\)) instead of joules. The electron volt is defined as the work done accelerating an electron through a potential difference of one volt, and the conversion to joules is as follows:
\(1\rm{eV}=1.6\times 10^{-19}J\)
Atomic emission spectra
Let’s see how the unique atomic energy levels of each element give rise to characteristic atomic emission spectra.
When an electron in hydrogen drops down from an excited state, such as \(n=2\) or \(n=3\), to the ground state, it emits a photon whose frequency depends on the energy difference of the transition:
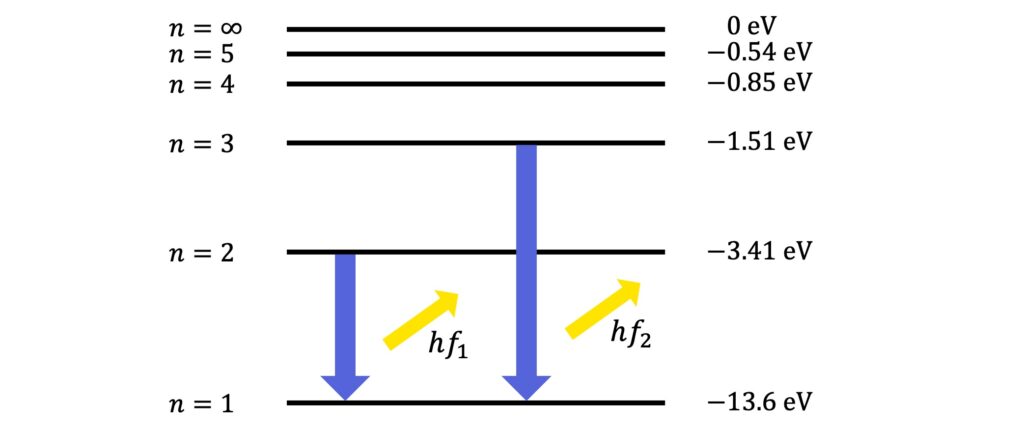
The frequencies generated by these transitions, as well as by those from higher levels, give rise to a characteristic set of frequencies emitted by hydrogen. This characteristic set of frequencies is the unique emission spectrum of hydrogen.
These frequencies form a subset of the continuous spectrum, the visible part of which looks like this:

The hydrogen emission spectrum can therefore be depicted as a subset of the continuous spectrum made up of emission lines at hydrogen’s characteristic frequencies:

Atomic absorption spectra
By contrast, atomic absorption spectra arise from the characteristic frequencies an element absorbs when its electrons transition to a higher level.
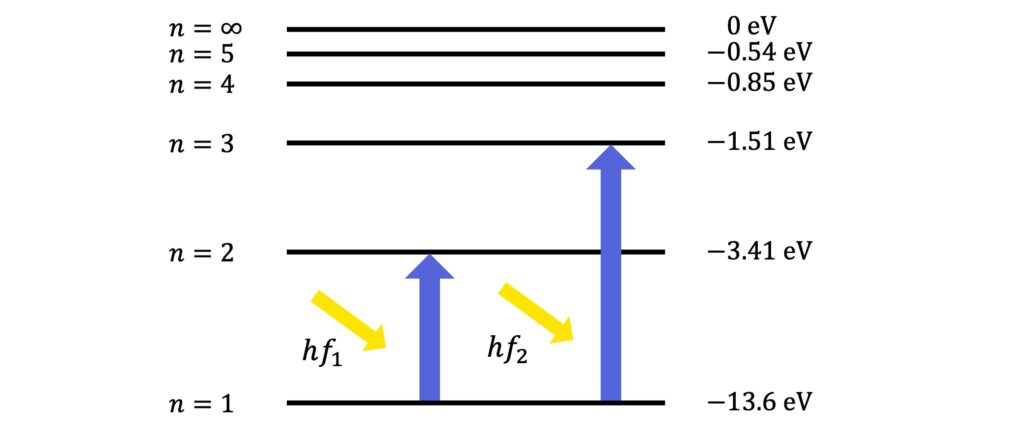
The characteristic absorption spectrum of hydrogen is as follows:

Unlike emission lines, absorption lines are shown as missing (black) parts of the continuous spectrum because these are the frequencies that are absorbed, or removed, from the continuous spectrum.
For any given element, the emission and absorption lines have the same frequencies because they depend on the differences between the energy levels of the element’s atoms.
An exception is that, while absorption usually takes place directly from the ground state, de-excitation can take place in multiple hops. For example, an electron in \(n=3\) can first transition to \(n=2\) and then subsequently to \(n=1\), releasing one photon for each hop. This produces additional emission lines compared to the absorption spectrum.
Atomic spectra in real-world applications
There is a wide range of practical applications of atomic spectra!
For example, neon lights contain an inert gas (such as neon or argon) inside a glass tube. A high voltage is placed across the gas which causes free electrons in the tube to accelerate, colliding with the atoms of the gas. This excites the atoms and ionises some of them by knocking their electrons free.
The continuous flow of free, high speed electrons causes the atoms and ions to frequently go into an excited state, and when their electrons drop back down to lower energy levels they release photons at characteristic frequencies. This is why neon lights shine with bright colours that are characteristic of the gas in the tube.
Fluorescent tubes are similar. They contain mercury vapour in a tube, across which a high voltage is applied. However, the de-excitation of the mercury atoms produces photons in the ultraviolet range (which are not visible). These photons land on a phosphor coating on the inside of the tube whose atoms excite from the ground state and then de-excite in a series of hops, releasing visible photons of various frequencies. This produces a more diffuse white light.
In other applications, the elements that are present in a sample can be identified by observing their atomic emission and absorption spectra. This approach has various applications, including enabling scientists to determine the elements present in the cooler outer atmospheres of stars by examining the absorption lines present in starlight.
Conclusion
I hope you’ve enjoyed this post on atomic energy levels and spectra! We’ve covered how each element has a unique set of atomic energy levels that gives rise to characteristic emission and absorption spectra. These phenomena have various practical and scientific applications.
If you’ve enjoyed this post, you might also like the related topic of the Doppler effect in starlight which builds directly on what you’ve learnt here today.
Happy studying!
